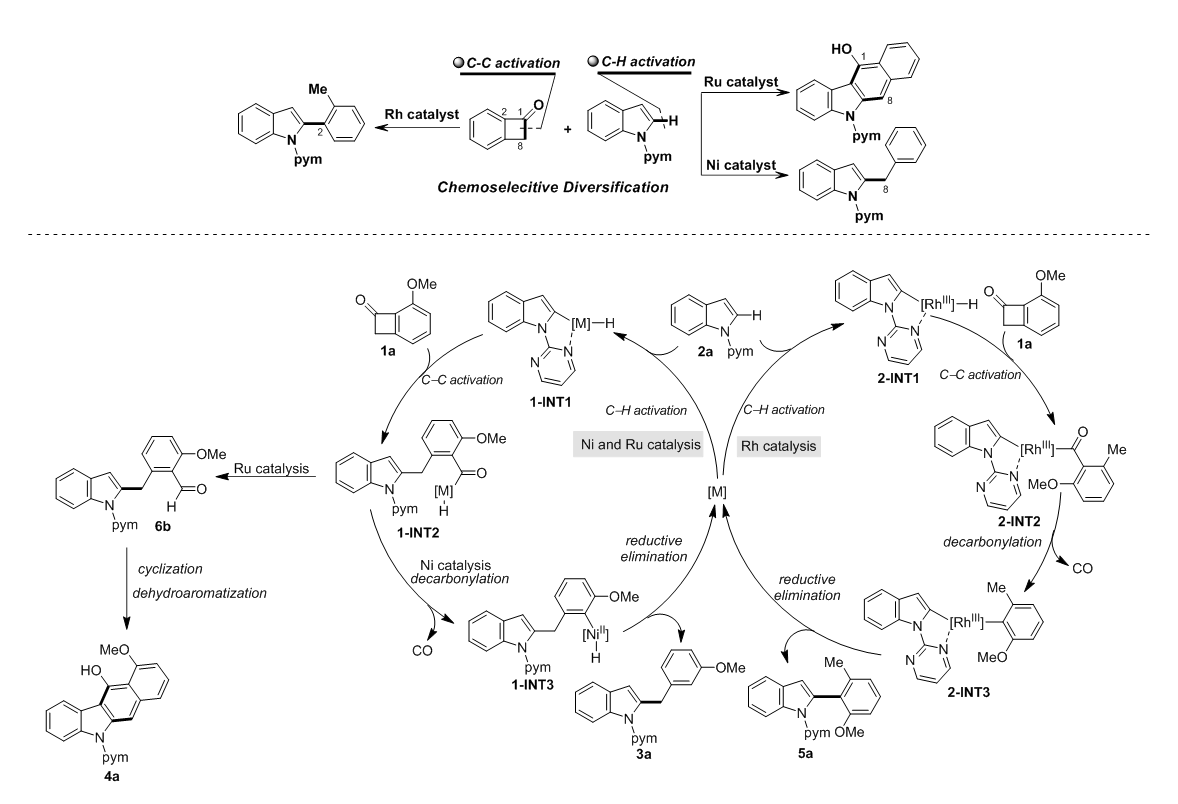
According to customs information, the European Commission has recently issued Notification G/TBT/N/EU/650, publishing a draft regulation setting limits on Hydroxyethoxyphenyl Butanone (HEPB) in cosmetics. The draft stipulates that when HEPB is used as a preservative in rinse-off, oral care, and leave-on cosmetic products, the maximum permitted concentration shall be 0.7%.
Records show that HEPB has long been used as a skin conditioning agent and was not listed in the prohibited or restricted substances under EU Cosmetics Regulation EC 1223/2009. However, as early as 2015, the European Commission received industry data confirming that this ingredient could also be used as a preservative in rinse-off, leave-on, and oral care products.
Subsequently, the European Scientific Committee on Consumer Safety (SCCS) evaluated the substance. The assessment concluded that HEPB is unsafe when used in rinse-off cosmetics, oral care products, or in leave-on cosmetics at concentrations above 2%. When limited to a maximum concentration of 0.7%, it is considered safe for use as a preservative in all relevant product categories.
Following multiple public consultations and reviews, the SCCS issued its final opinion on March 11, 2019, regarding the safety of HEPB in cosmetics: when added as a preservative to oral care rinses, the maximum concentration must not exceed 0.7%, as higher concentrations may cause eye irritation.
The European Commission then formally confirmed this limit in Notification G/TBT/N/EU/650: the maximum concentration of HEPB as a preservative in oral care rinses is set at 0.7%.
Since HEPB can also function as a skin conditioning agent, current EU regulations require that if a multi-functional cosmetic ingredient is approved for use as a preservative, its concentration for all other applications must not exceed the maximum limit set for preservative use. Enterprises exporting to the EU are advised to review their formulations promptly and make necessary adjustments.